Lead Indication
Metabolic dysfunction-associated steatohepatitis (MASH)
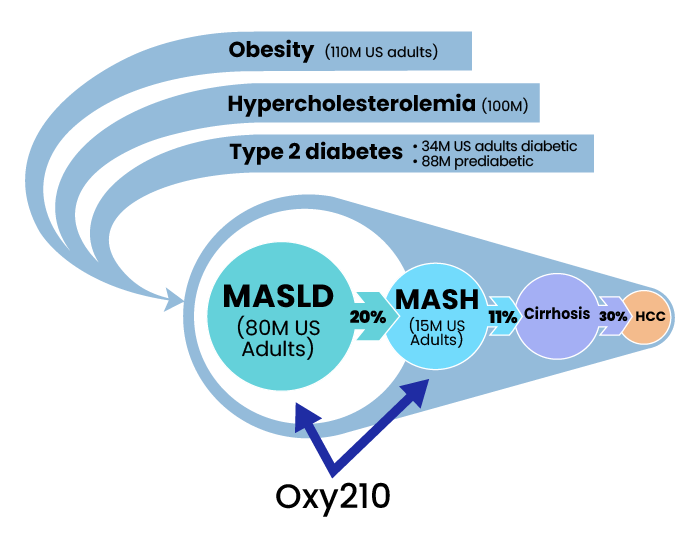
Millions of American adults suffer from obesity, metabolic syndrome, diabetes and cardiovascular disease associated with hyperlipidemia. About 110 million adults in the US are obese; about 88 million, or 34.5% of American adults, are prediabetic; and about 34.2 million Americans suffer from type-2 diabetes. Among this huge patient population, Metabolic dysfunction-associated steatotic liver disease (MASLD), formerly known as non-alcoholic fatty liver disease (NAFLD) is very common. In fact, about 80 million, or 30% of the US adult population, suffer from MASLD. Globally, between 20 to 30 percent of the populations in Western developed countries are affected by MASLD. While MASLD can often be benign and reversible with improvements in diet and lifestyle, about 20% of all MASLD patients are at increased risk for developing MASH, a more dangerous inflammatory liver disease that can progress toward liver cirrhosis and liver cancer. Currently, MASH affects an estimated 15 million American adults, and this number is expected to rise exponentially in the coming years, driven by the increasing prevalence of obesity, diabetes and metabolic syndrome. The global MASH market size is projected to reach $31 billion by 2027, from $2 billion in 2020, at a CAGR of 46.7% during 2021-2027. In 2017, the direct lifetime costs associated with MASLD/MASH were $222.6 billion; $95.4 billion of this was for patients with MASH.
It is estimated that by 2030, complications resulting from MASH will be the leading cause for liver transplantation procedures in the US. Currently, Rezdiffra is the only FDA approved therapy developed specifically for MASH, is effective in less than 30% of MASH patient population and only indicated for those with non-cirrhotic MASH. More recently, the FDA approved GLP-1 agonist Wegovy (semaglutide) as a therapy for MASH that works mainly through inducing weight loss. Changes in diet and lifestyle and the use of some off-label drugs (e.g., statins) can also help support MASH patients, depending on the stage of the disease.
Over the last decades, the development of therapies for MASH has become a high priority of the pharmaceutical industry and a diverse pipeline of innovative drug candidates has been progressing toward the clinic in recent years. However, due to the complex nature of the disease and pipeline attrition, drug failures during late-stage clinical trials have been even more common than in other therapeutic areas, such as cancer, for example. The causes for MASH are multifactorial and incompletely understood, and it seems likely that combination therapies do not result in better clinical outcomes than monotherapies, adding additional layers of complication to clinical trial design.
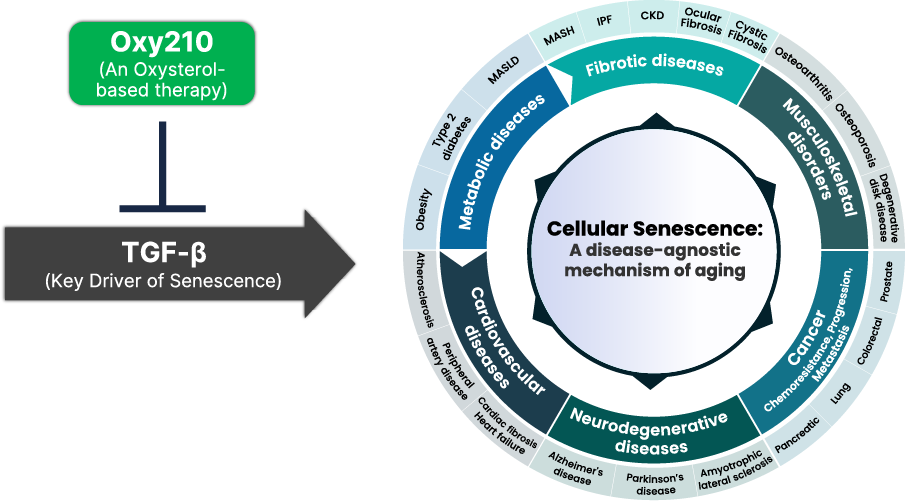
Through our drug discovery platform Oxysterol Therapeutics® technology, we have identified Oxy210, a proprietary oxysterol drug candidate, for targeting MASH. Oxy210 displays a unique combination of anti-inflammatory and antifibrotic properties driven in part by inhibiting three major drivers of MASH: 1) transforming growth factor-beta (TGF-β) signaling in hepatic stellate cells (HSC; cells that are responsible for fibrosis in the liver), 2) Hedgehog (Hh) signaling in HSC that causes their activation, and 3) Toll-Like Receptor (TLR) signaling in Kupffer cells and macrophages that are responsible for inflammation in the liver. In a preclinical humanized hyperlipidemic mouse model of MASH, Oxy210 significantly inhibited liver inflammation and fibrosis and showed promising disease modifying effects, in addition to favorable drug safety and pharmacokinetic (PK) profiles. Importantly, given that the majority of patients with MASH die of cardiovascular complications, Oxy210 also inhibited atherosclerosis that develops in parallel with MASH through its cholesterol lowering and anti-inflammatory effects.
Also significant is our recent finding that Oxy210, in addition to its antifibrotic and anti-inflammatory effects, inhibits cellular aging (senescence) in vitro and in vivo in preclinical models of fibrotic diseases, including MASH and idiopathic pulmonary fibrosis (IPF). With the greater emphasis being placed by the medical and scientific community on longevity and aging research, this has further brought importance to Oxy210 as a potential drug candidate.
Why Our Work is Important
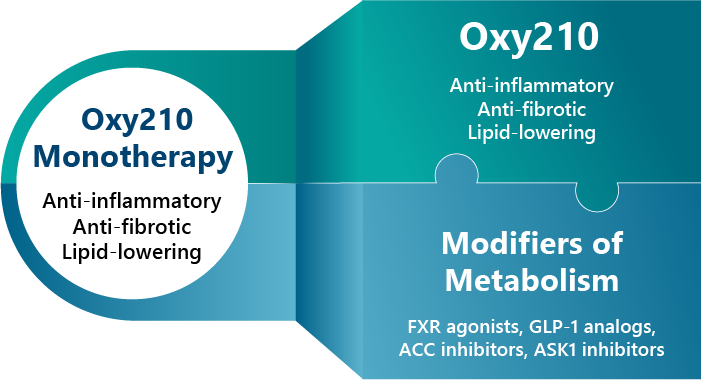
We believe that Oxy210 could be developed into a safe and effective treatment for MASLD and NASH patients, as well as individuals with a predisposition to developing these conditions, for example, patients with diabetes and obesity. Oxy210 is orally bioavailable and can be inexpensively produced on scale. Oxy210 is naturally sequestered by the liver after oral administration due to its oxysterol-based properties. Furthermore, Oxy210 may be used as a monotherapy or in combination with other drug candidates that are in the pipeline of other biopharmaceutical companies, for example, farnesoid X receptor (FXR) activators, stearoyl‐CoA desaturase‐1 (SCD-1) modulators, FGF-21 analogues and Glucagon-like peptide-1 (GLP-1) inhibitors. In March 2024 the FDA approved Rezdiffra (resmetirom), an agonist of thyroid hormone receptor-beta (THR-β), indicated for the treatment of non-cirrhotic MASH. In clinical trials, Rezdiffra showed efficacy in less than 30% of patients. In August 2025 Wegovy (semaglutide) was also approved by the FDA for non-cirrhotic MASH in spite of modest efficacy. By targeting multiple cellular and molecular drivers of MASH, we believe that due to its multi-specificity, Oxy210 will distinguish itself from other MASH drug candidates that are specifically designed to affect only a single target.
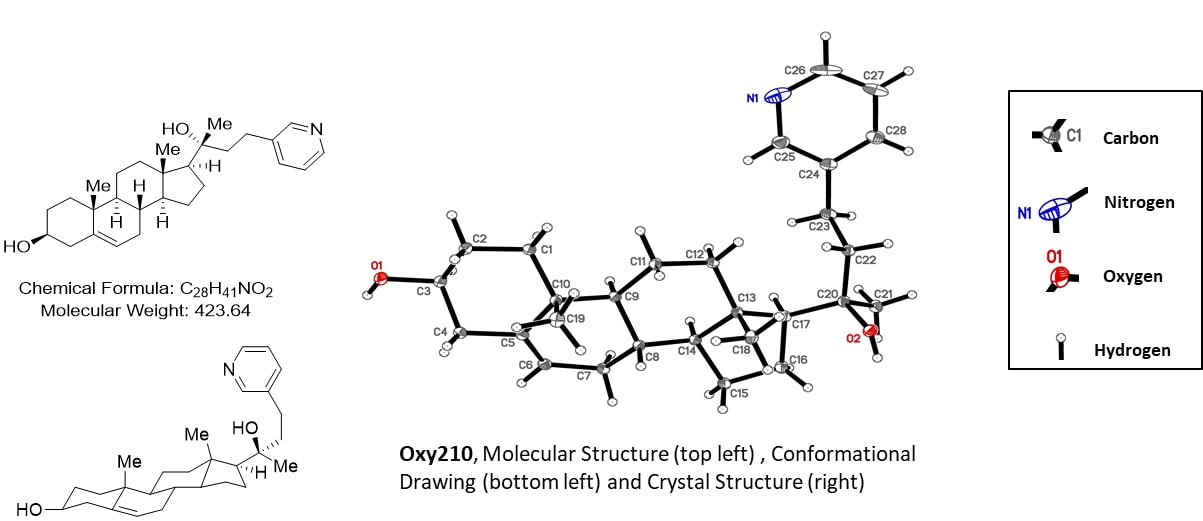
Oxy210
Preclinical Pharmacology
Oxy210 is a semi-synthetic oxysterol displaying a unique combination of antifibrotic and anti-inflammatory properties. Oxy210 is derived from a naturally occurring oxysterol, 20α,22(R)-dihydroxy-cholesterol (Oxy16), a known metabolite of cholesterol in the biosynthesis of steroid hormones, which is also a weak antagonist of Hedgehog signaling. Oxy210 can be prepared in a three-step chemical synthesis, using inexpensive and readily available naturally occurring pregnenolone as a starting material. Furthermore, Oxy210 significantly reduces cholesterol levels in circulation as well as extrahepatic systemic inflammation that occurs in parallel with the development of MASH and obesity in preclinical models.
Mechanism of Action
The antifibrotic activity of Oxy210 is mediated by inhibition of two pro-fibrotic signaling pathways in hepatic stellate cells (HSCs), namely Hedgehog and TGF-β signaling. In addition, Oxy210 possesses anti-inflammatory activity in part through inhibition of Toll-Like Receptor 2 (TLR2) and TLR4, independent of cyclooxygenase 1 (COX-1), COX-2 or steroid receptor inhibition. Oxy210 also inhibits senescence (cellular aging) in cultured hepatocytes and in the liver that contributes to exacerbation of inflammation and fibrosis in MASH livers.
Safety
Oxy210 is well tolerated in mice with acute and chronic dosing. In one recent study, Oxy210, mixed into mouse chow at a loading of 4 mg/gram of food, was well tolerated in mice for 16 weeks of chronic dosing. This corresponds to a dose of about 260 mg/kg/day (based on food consumption). Oxy210 administered at high doses to pregnant mice did not cause harm to embryonic development and hence is non-teratogenic.
Oxy210 did not inhibit hERG ion channel function, hence deemed safe to cardiac system, and tested negative in the Ames mutagenicity assay, suggesting lack of genetic toxic effects.
Safety target and off-target screening and paneling showed no significant agonist or antagonist activity toward G protein-coupled receptors, nuclear hormone receptors, and ion channels.
Efficacy
Using a disease relevant “humanized” MASH mouse model, Oxy210 was shown to effectively inhibit hepatic fibrosis and inflammation and improve hypercholesterolemia. Oral administration of Oxy210 results in inhibition of hepatic inflammatory and fibrotic gene expression, reduced lipid deposition and apoptosis, senescence, improved hepatic function, reduced hepatic fibrosis associated with significant inhibition of other MASH hallmarks. The antifibrotic and anti-inflammatory effects of Oxy210 are at least in part mediated through inhibition of three known drivers of MASH: Hedgehog signaling, TGF-β signaling and Toll-like receptor signaling. In addition to its effect on MASH, Oxy210 also inhibits atherosclerosis and adipose tissue inflammation that occur in parallel with the development of MASH. In more recent studies, Oxy210 was also found to inhibit high fat diet-induced heart failure in a preclinical mouse model.